Dissolved oxygen (DO) is a vital parameter for assessing water quality, supporting aquatic life, and ensuring efficient industrial processes. A dissolved oxygen sensor, or DO sensor, is a specialized device designed to measure the concentration of oxygen gas dissolved in water. From aquaculture farms to wastewater treatment plants, DO sensors play a pivotal role in maintaining healthy ecosystems and optimizing operations. This article explains the function of a DO sensor, how it works, and its diverse applications.

Dissolved oxygen is essential for:
- Aquatic Life: Fish, invertebrates, and microorganisms rely on oxygen for survival.
- Water Quality: Low DO levels indicate pollution, eutrophication, or algal blooms.
- Industrial Efficiency: Aerobic bacteria in wastewater treatment require oxygen to break down organic matter.
- Environmental Research: Scientists study DO to monitor ecosystem health and climate change impacts.
A DO sensor provides real-time data to prevent hypoxia (oxygen depletion) and ensure compliance with regulatory standards.
A DO sensor measures the amount of oxygen dissolved in water, typically expressed in milligrams per liter (mg/L) or percentage saturation (%Sat). Its primary functions include:
1. Quantifying Oxygen Levels: Detects hypoxic or anoxic conditions that threaten aquatic organisms.
2. Monitoring Water Treatment: Ensures optimal oxygen levels for aerobic digestion in wastewater plants.
3. Supporting Research: Tracks seasonal or diurnal oxygen fluctuations in natural water bodies.
4. Preventing Equipment Damage: Controls DO levels in boilers and cooling systems to minimize corrosion.
DO sensors operate using two main technologies: **electrochemical** and **optical** sensing.
1. Electrochemical DO Sensors
- Principle: These sensors use an electrochemical cell with a cathode, anode, and electrolyte solution.
- Process:
- Oxygen diffuses through a gas-permeable membrane into the electrolyte.
- At the cathode, oxygen undergoes reduction, generating an electric current proportional to DO concentration.
- The sensor converts this current into a DO reading.
- Types:
- Galvanic Sensors: Self-powered, with a zinc anode and silver cathode.
- Polarographic Sensors: Require external voltage to polarize the electrodes.
- Advantages: Cost-effective, long-lasting, and widely used in industrial settings.
- Drawbacks: Require regular membrane and electrolyte replacements.
2. Optical DO Sensors
- Principle: These sensors use luminescent technology with a light-sensitive dye.
- Process:
- A blue light excites the dye, causing it to emit red light.
- Oxygen molecules interact with the dye, quenching (reducing) the light intensity and duration.
- The sensor measures this quenching effect to calculate DO levels.
- Advantages: Minimal maintenance, no electrolyte, and resistance to flow rate changes.
- Drawbacks: Higher initial cost and sensitivity to fouling.

1. Aquaculture and Fish Farming:
- Maintains optimal DO levels to prevent fish stress and mortality.
- Monifies water quality in shrimp ponds and hatcheries.
2. Wastewater Treatment:
- Controls aeration systems to ensure efficient breakdown of organic waste.
- Prevents over-aeration, reducing energy costs.
3. Environmental Monitoring:
- Tracks DO in rivers, lakes, and oceans to assess ecosystem health.
- Identifies dead zones caused by nutrient pollution.
4. Industrial Processes:
- Monitors DO in boilers to prevent corrosion and scale formation.
- Ensures compliance in food and beverage production (e.g., breweries).
5. Research and Education:
- Studies oxygen dynamics in climate change models.
- Teaches students about aquatic biology and chemistry.
1. Calibration:
- Calibrate the sensor using a zero-oxygen solution (sodium sulfite) and air-saturated water.
- Follow manufacturer guidelines for temperature and pressure compensation.
2. Sample Preparation:
- Collect water in a clean container, avoiding air bubbles or agitation.
- For stagnant water, gently stir the sample to mimic natural flow.
3. Measurement:
- Submerge the sensor probe fully and wait for readings to stabilize (1–2 minutes).
- Record DO levels alongside temperature and salinity for comprehensive analysis.
4. Maintenance:
- Clean the membrane or optical cap regularly to remove biofilm or debris.
- Store electrochemical sensors in a moist environment to prevent membrane drying.
- Temperature: DO solubility decreases as water warms. Most sensors auto-compensate for temperature.
- Salinity: Saltwater holds less oxygen than freshwater. Use sensors with salinity correction.
- Atmospheric Pressure: High-altitude locations require pressure compensation for accurate %Sat readings.
- Biofouling: Algae or bacteria on the sensor surface can skew results.
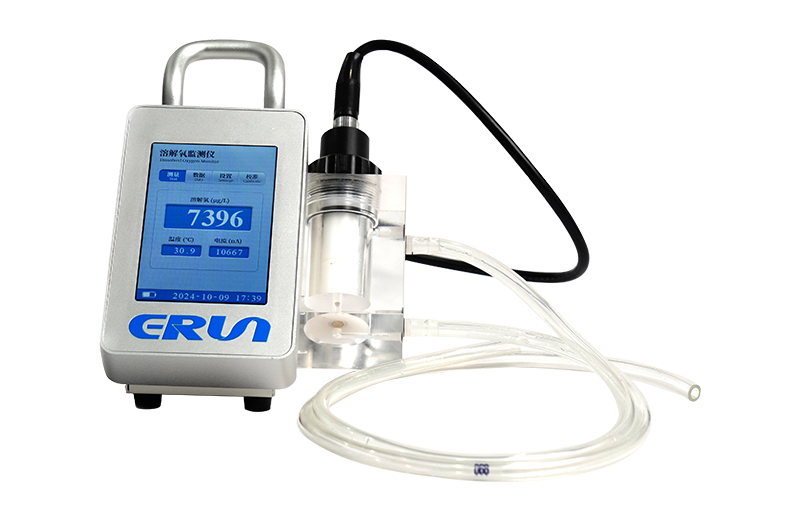
- Application: Select electrochemical sensors for cost-sensitive industrial use and optical sensors for low-maintenance fieldwork.
- Accuracy: Opt for ±0.1 mg/L precision for critical environments like fish farms.
- Durability: Choose titanium or stainless-steel probes for corrosive or marine environments.
- Features: Look for built-in data logging, Bluetooth connectivity, or multi-parameter capabilities.
A DO sensor is more than a tool—it’s a guardian of water quality. By providing precise oxygen measurements, it helps industries protect aquatic life, optimize processes, and comply with environmental regulations. Whether you’re managing a wastewater plant, studying oceanography, or running a fish farm, understanding the function of a DO sensor empowers you to make informed decisions and sustain healthy water ecosystems.