Dissolved Oxygen (DO) is not just a parameter on a report; it is the lifeblood of any aquatic ecosystem and a non-negotiable variable in industrial water treatment. The question “how does dissolved oxygen affect water quality” is foundational for environmental scientists, aquaculture managers, and process engineers. Whether you are dealing with a fragile marine habitat or a high-intensity wastewater aeration basin, DO concentration dictates the biological and chemical stability of the water.
Understanding this dynamic is essential because low DO is a leading indicator of pollution and ecosystem distress, while properly managed DO levels ensure compliance with EPA and international water quality standards. This article explores the science behind DO, its critical impact on water quality, and the technology required to measure it accurately in real-time.
Dissolved oxygen refers to the amount of gaseous oxygen (O₂) dissolved in water. Unlike the oxygen in the air we breathe, DO enters water primarily through two pathways:
Direct atmospheric absorption, enhanced by turbulence such as waves or waterfalls
As a byproduct of photosynthesis from aquatic plants and algae
The oxygen-holding capacity (saturation) of water is not constant. It is inversely related to water temperature and salinity: cold freshwater holds significantly more oxygen than warm saltwater. This relationship explains why climate change and thermal pollution pose severe threats to aquatic life.
DO concentration acts as a barometer for water quality. Its effects are categorized below:
Most aerobic aquatic organisms require a minimum of 4–5 mg/L DO to survive. Below this threshold:
Hypoxia (Low Oxygen): Organisms suffer stress, reduced growth, and higher disease susceptibility
Anoxia (No Oxygen): DO at 0 mg/L creates “dead zones” with massive fish kills and ecosystem collapse
Species Shift: Low DO favors pollution-tolerant species (e.g., sludge worms) over sensitive species (e.g., trout, salmon), reducing biodiversity
Regulatory minimums:
Warm-water systems: ≥ 5.0 mg/L
Cold-water systems: ≥ 6.0 mg/L
DO controls key chemical reactions in water:
Oxidation of Metals: High DO precipitates iron and manganese (causing staining); low DO keeps them dissolved and mobile
Decomposition: Aerobic bacteria consume DO to break down organic waste (sewage, agricultural runoff). Excessively high Biological Oxygen Demand (BOD) collapses DO levels, producing septic, foul-smelling water. Clean waters require BOD₅ < 3 mg/L
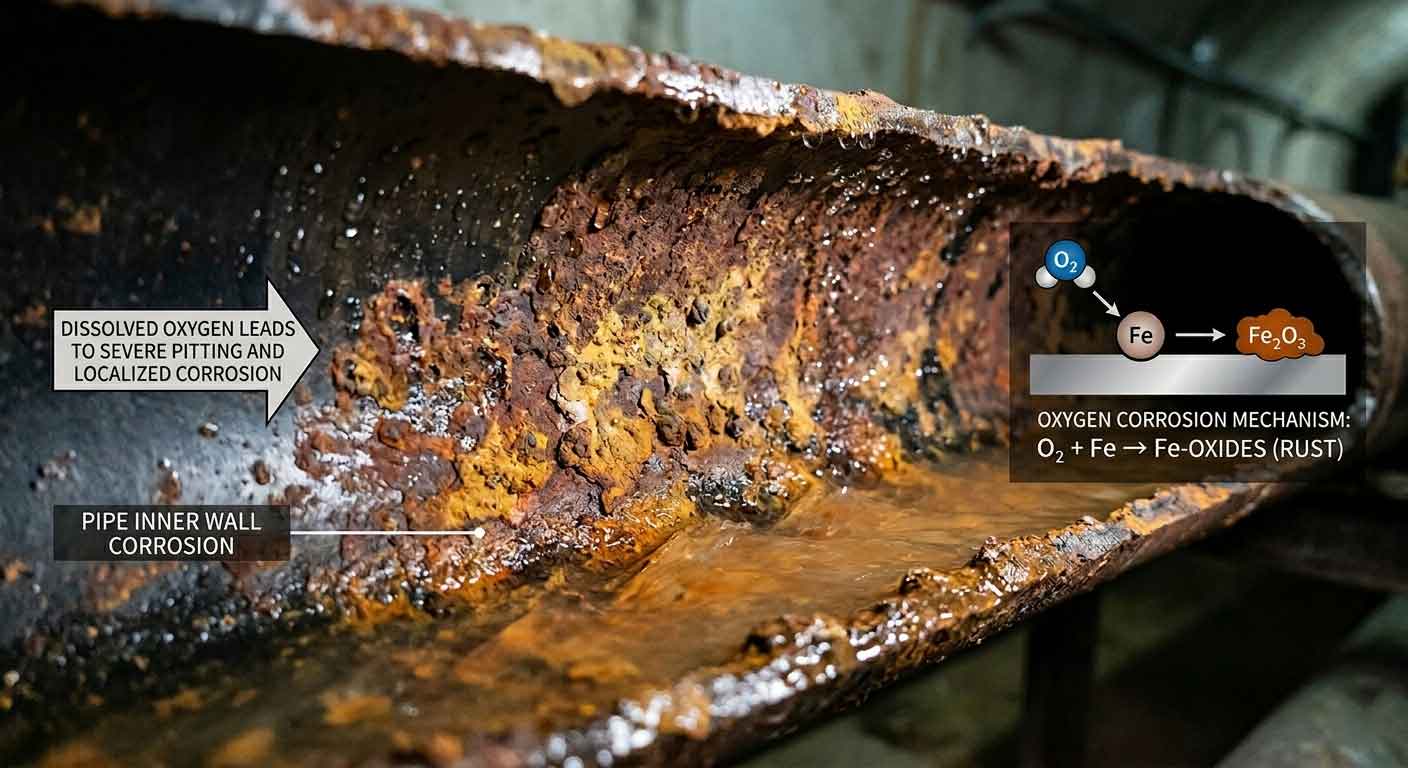
DO is not a primary human health concern in finished drinking water but strongly affects corrosion:
High DO accelerates corrosion of iron and steel pipes, causing “red water” and infrastructure damage
Low DO in raw water pipelines promotes biofilm growth and pre-treatment water quality deterioration
Major drivers of DO variation include:
Temperature: Warmer water holds less oxygen; seasonal changes and thermal discharge reduce DO
Salinity: Higher salinity lowers oxygen solubility
Organic Pollution: Sewage and runoff boost bacterial oxygen consumption beyond replenishment
Diurnal Cycle: Algae‑rich waters show DO supersaturation by day (photosynthesis) and critical lows at night (respiration)
Laboratory sampling is often insufficient. Continuous online monitoring is essential for aquaculture and wastewater treatment. Two dominant sensor technologies are compared below:
Feature | Electrochemical (Clark Cell) Sensors | Optical (Luminescence) Sensors |
Principle | Oxygen diffuses through a membrane; chemical reaction generates current | Oxygen quenches dye fluorescence; measured optically |
Maintenance | High: regular electrolyte replacement and membrane cleaning | Low: no electrolyte; minimal drift |
Flow Dependency | Yes: requires constant flow for accuracy | No: works in static or flowing water |
Interferences | Susceptible to H₂S and membrane poisoning | Unaffected: ideal for harsh environments |
Accuracy | Good, but drifts with fouling | Excellent long‑term stability |
Optical DO sensors are the modern gold standard for low maintenance and high reliability.

The ERUN‑A5F Online Intelligent Dissolved Oxygen Sensor provides robust, low‑maintenance monitoring for industrial and environmental applications.
Advanced Optical Technology: Fluorescence quenching, no oxygen consumption, accurate in low‑flow conditions
Wide Range: 0.00 – 20.00 mg/L, resolution 0.01 mg/L, suitable for freshwater and seawater
Durable Design: IP68 waterproof, rugged ABS housing for continuous immersion
Easy Integration: RS485 (Modbus‑RTU) and 4–20mA output for SCADA, PLC, and IoT platforms
Low Maintenance: No frequent electrolyte or membrane changes, stable long‑term calibration
Ideal for fish farms, wastewater discharge monitoring, and coastal water quality protection.
Dissolved oxygen is the definitive measure of an aquatic system’s ability to sustain life and process waste. Mastering how dissolved oxygen affects water quality helps prevent fish kills, optimize treatment efficiency, and meet strict environmental regulations.
With advanced optical monitoring tools such as the ERUN‑A5F Online Intelligent DO Sensor, water professionals can shift from reactive troubleshooting to proactive management, safeguarding water resources for long‑term health and productivity.